|
feature the company's proprietary, low-energy BurstDR waveform. 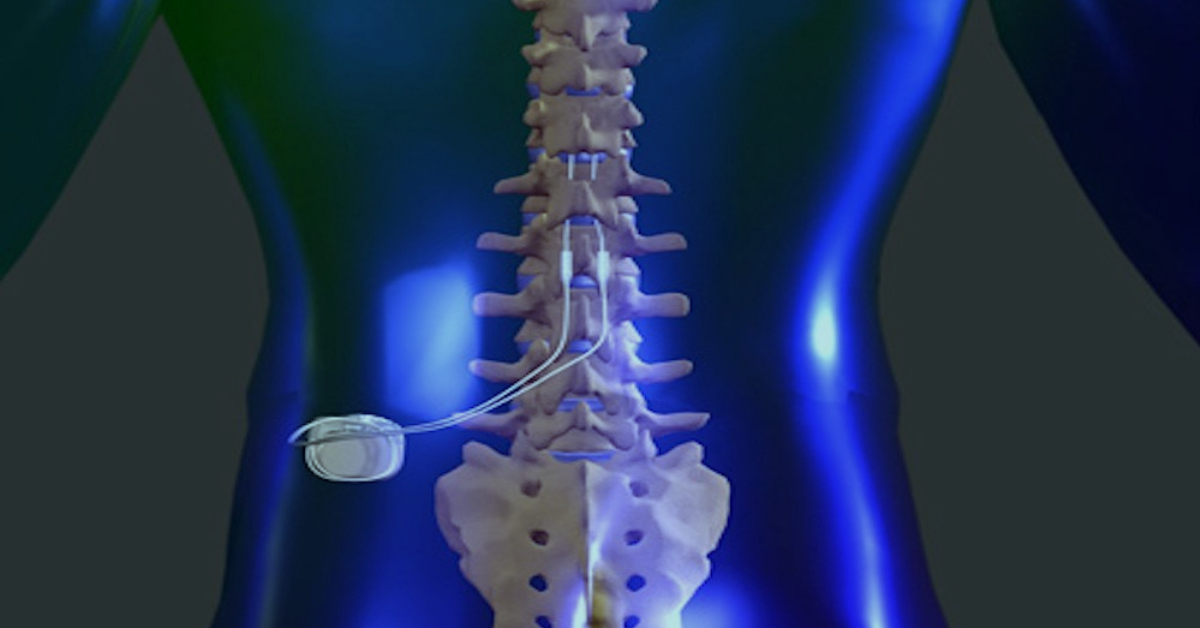 
All of Abbott's SCS therapies in the U.S. This new indication will span across the entirety of Abbott's SCS portfolio in the U.S., which includes the recharge-free Proclaim SCS family and the rechargeable Eterna SCS platform. Results at six months for the first 200 patients demonstrated clear clinical evidence supporting BurstDR stimulation therapy as an effective treatment for chronic back pain and were sustained at the 12-month follow-up. This labeling expansion was supported by results from the DISTINCT study, which demonstrated that Abbott's proprietary BurstDR SCS technology improved pain levels, ability to perform daily activities and emotional well-being in people with chronic back pain.Ībbott's DISTINCT study, which enrolled 270 participants who suffered for an average of 12.8 years with pain, is the largest randomized controlled trial for SCS in people with chronic back pain when surgery is not an option. 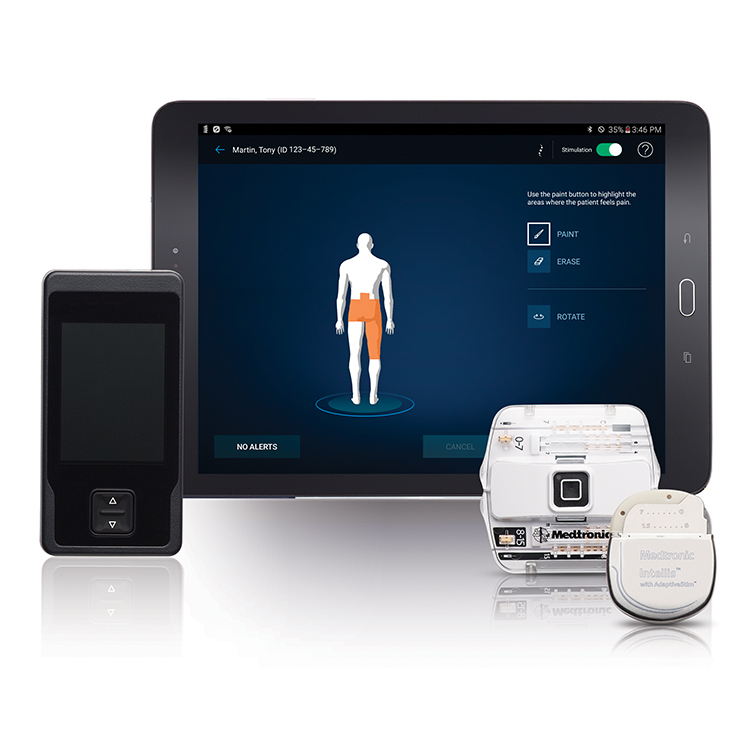
Food and Drug Administration (FDA) has approved its spinal cord stimulation (SCS) devices for the treatment of chronic back pain in people who have not had or are not eligible to receive back surgery, known as non-surgical back pain. Walgreens settles New York charges it grossly inflated prices of infant formulaĪbbott Laboratories Recalling HeartMate Touch Communication SystemĪbbott Laboratories Says Real-World Data Show GLP-1 Medicines With FreeStyle Libre Help Manage Type 2 DiabetesĪbbott Announces Real-World Data Show Freestyle Libre Systems and GLP-1 Medicines Work Better Together for People with Type 2 DiabetesĪbbVie CEO to Retire COO Robert Michael Named SuccessorĪbbVie Names Robert Michael to Succeed Richard Gonzalez as CEOĪbbott Laboratories Maintains Quarterly Dividend at $0.55 a Share, Payable May 15 to Holders of Record April 15.(RTTNews) - Abbott Laboratories (ABT) announced Tuesday that the U.S. Reckitt unit hit with $60 million verdict in Enfamil baby formula case in Illinois Reckitt shares sink as investors see more trouble from baby formula lawsuits US FDA approves Abbott's heart valve repair deviceĪbbott Receives FDA Approval for Triclip?, First-Of-Its-Kind Device to Repair Leaky Tricuspid Heart ValveĪbbott Laboratories Says Its i-STAT TBI Cartridge Received US FDA Clearance for Use With Whole BloodĪbbott Laboratories Cartridge Gets FDA Clearance to be Used with Whole BloodĪbbott Receives FDA Clearance for Whole Blood Rapid Test to Help with Assessment of Concussion at the Patient's BedsideĪbbott Laboratories' Assert-IQ Insertable Cardiac Monitor Gets CE Mark in EuropeĪbbott Advances Chronic Pain Management with Small Implanted, Rechargeable Spinal Cord Stimulation System S&P 500 Posts Second Consecutive Weekly Drop Led by Financials Amid Higher-Than-Expected Consumer PricesĬitigroup Raises Price Target on Abbott Laboratories to $128 From $126, Keeps Buy RatingĪbbott's heart valve repair device gets US approvalĪbbott Laboratories' TriClip Receives FDA Approval for Tricuspid Valve RepairĪbbott Gets FDA Approval for TriClip Heart Valve Repair System The Eterna SCS System is currently the small device available, designed for implant site comfort, and features Abbott's BurstDR stimulation, which mimics natural firing patterns found in the brain to deliver superior pain relief over traditional tonic stimulation. This Medicare coding and payment guide offers information about reimbursement for Spinal Cord Stimulation (SCS) and Dorsal Root Ganglion Stimulation (DRG). The studies found that people wanted small, comfortable implants which don't need to be charged daily.Ībbott designed the Eterna SCS System to be recharged wirelessly at a frequency of five times per year under normal use, making it the lowest recharge burden platform on the market. use only HE&R approved for non -promotional use only. 
Approved by Health Canada in 2023, Abbott developed the Eterna SCS System based on extensive studies with patients, physicians, and caregivers to understand the unmet needs of people living with chronic pain. The Eterna SCS system uses Abbott's proprietary BurstDR stimulation, the only SCS technology with the high level of clinical evidence (level 1A.) Abbott's proprietary BurstDR therapy is proven to reduce pain by 23% more than traditional tonic stimulation without the tingling sensation. The inaugural use case of the Eterna SCS system took place at Hôpital de l'Enfant-Jésus hospital in Quebec City. Abbott announced that the company's Eterna SCS system ? the small implantable, rechargeable spinal cord stimulator (SCS) for the treatment of chronic pain, has been used for the first time in Canada.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
